Could Newly Found “Peacekeeping” Cells Be a Weapon against COVID-19?
To struggle a respiratory infection, the body requires a two-pronged attack. Very first, it sends immune cells to the scene to demolish the pathogen. Then the defense program must continue to keep all those initial responders from spiraling out of handle. If this attempt at “peacekeeping” fails, a run-of-the-mill fever and cough can escalate to a life-threatening illness—which transpired to the tens of thousands of COVID-19 sufferers who have succumbed to the global pandemic caused by the SARS-CoV-2 virus.
For the most element, macrophages—the significant immune cells that consume pathogens—are initial responders. In the lungs of mice infected with viral influenza, even so, a little subset of these white blood cells does just the opposite: They suppress surplus swelling, scientists report these days in the journal Science Immunology. These peacekeeping macrophages also reside in human lungs, suggesting they “might be quite essential to support COVID-19 sufferers resist swelling and possibly survive,” says immunologist Yufang Shi of the Very first Affiliated Clinic of Soochow College in China. The clinic despatched personnel and supplies to the nation’s city of Wuhan, but Shi was not included in the new research.
The analysis started seven yrs back, when Kamal Khanna, an immunologist now at NYU Langone Health and fitness, seen one thing he discovered to be spectacular. At the time, his lab was finding out a related group of macrophages—not in the lungs but in the spleen, a blood-filtering organ in the lymphatic program. On stained mouse tissue considered less than a microscope, the macrophages formed blue rings around immune-cell-wealthy regions of the spleen. “They looked like nebulas,” Khanna says.
And these cells ended up not just visually spectacular. When the scientists depleted the macrophages applying a clever genetic strategy, the mice died just two days following currently being infected with little quantities of Listeria microbes they would commonly obvious. Yet another observation was also placing: although other immune cells packed infection-combating zones in the spleen, this group of macrophages stayed set. “And we imagined, This compartmentalization has to be existing in [nonimmune] organs as nicely,” Khanna says. The spleen conclusions, revealed in 2017, laid the groundwork for the new investigation in lungs.
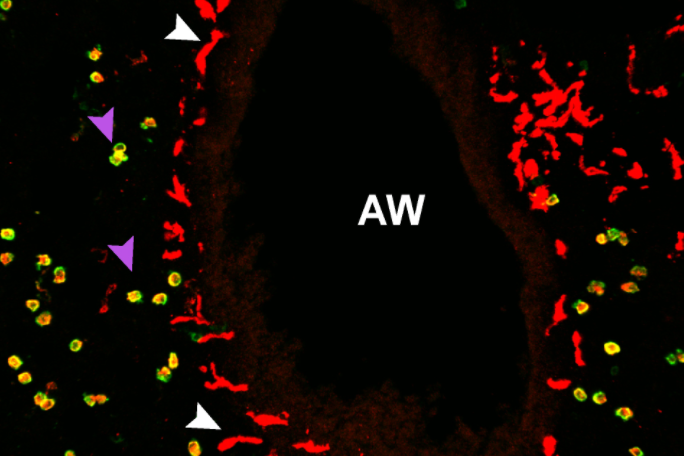
In this sophisticated organ, the vast the greater part of macrophages are living in little air sacs referred to as alveoli. But when the scientists examined the lung tissue less than the microscope, they observed a substantially scaled-down population that was starkly distinct. Unlike alveolar macrophages (AMs), which are significant and round, the rarer macrophages are elongated with sprawling arms — and they’re not discovered in alveoli. Identified as nerve- and airway-affiliated macrophages, or NAMs, these newly identified cells congregate at airways and interact with bordering nerves. “The full airway branch gets lit up with these macrophages,” Khanna says.
In one more established of reports, his crew depleted mice of AMs or NAMs and then infected these animals and standard mice with an influenza virus and as opposed the level of virus in the groups. These experiments uncovered a division of labor: AMs support to struggle the virus although NAMs continue to keep the peace and avert tissue destruction.
This type of differentiation could demonstrate essential for designing therapies focused at swelling, which is a large difficulty in COVID-19, says Mallar Bhattacharya, a macrophage biologist at the College of California, San Francisco, who was not included with the analysis but calls it a “clever software of novel equipment for deletion of particular macrophage subsets.”
NAM-depleted mice created bigger stages of numerous inflammatory molecules, including a person referred to as IL-six that is included in so-referred to as “cytokine storms” noticed in some sufferers with severe COVID-19. In a recent research of 191 folks handled for the sickness in Wuhan, blood IL-six stages ended up elevated in sufferers who died of it, as opposed with survivors. Clinical trials are now assessing IL-6–blocking antibodies—drugs that are made use of to take care of rheumatoid arthritis—in COVID-19 sufferers.
The new research did not address how the intertwining of NAMs with nerves relates to the function of these immune cells. Khanna hopes to achieve perception in long run mouse reports by depleting NAMs and evaluating the health and fitness of bordering nerves or by inspecting how the airway nerves are afflicted through distinct sorts of infections. The anxious-immune relationship is intriguing in mild of recent analysis suggesting that chemical cross communicate amongst gut macrophages and nerve fibers can handle peristalsis, the course of action that moves food items via the digestive tract.
A far more urgent problem is whether or not NAMs are included in COVID-19. Towards that close, Khanna is functioning with NYU Langone Health and fitness to receive new lung tissue from folks who died of the disease—but accomplishing so is logistically difficult and quite possibly dangerous. An even larger problem for now, in mild of New York City’s rising amount of conditions, is that “basically, our lab is shut down,” Khanna says.
Go through far more about the coronavirus outbreak below.




